BISAC NAT010000 Ecology
BISAC NAT045050 Ecosystems & Habitats / Coastal Regions & Shorelines
BISAC NAT025000 Ecosystems & Habitats / Oceans & Seas
BISAC NAT045030 Ecosystems & Habitats / Polar Regions
BISAC SCI081000 Earth Sciences / Hydrology
BISAC SCI092000 Global Warming & Climate Change
BISAC SCI020000 Life Sciences / Ecology
BISAC SCI039000 Life Sciences / Marine Biology
BISAC SOC053000 Regional Studies
BISAC TEC060000 Marine & Naval
A brief overview of the coastal biodiversity of Vietnam based on surveys conducted by the A.V. Zhirmunsky Institute of Marine Biology, Far Eastern Branch of the Russian Academy of Sciences for last 35 years. Main problems related to threats to biodiversity are discussed on the example of the intertidal communities, coral reefs, and molluscan diversity. Threats to marine biodiversity in Vietnam are summarized as follows: habitat degradation, fragmentation and loss (especially important are mangrove forest destruction, loss of coral reefs, change in landscape mosaic of wetland, estuary, sand and mud flats); global climate change including sea level rise, storm events, rainfall pattern change, warming of the coastal ocean; effects of fishing and other forms of overexploitation; pollution and marine litter; species introduction/invasions; physical alterations of coasts; tourism. Consolidated data of Vietnamese and Russian researchers on biodiversity and coastal zone management can be used in interpretations of ecosystem changes and for development of recommendations for local/national decision-makers.
Vietnam, marine biodiversity, status, threats, assessment.
I. Introduction
The South China Sea, including its Vietnamese coast, is one of the richest regions of the World Ocean in terms of biodiversity. The sea is partially included into the East Indies Triangle, or the Coral Triangle [1], a centre of maximum marine biodiversity. The A.V. Zhirmunsky Institute of Marine Biology, Far Eastern Branch of the Russian Academy of Sciences (Vladivostok; hereafter, the IMB) has been studying the marine biological diversity in coastal zones of Vietnam since the early 1980s, its status, threats, recent modifications due to global change and human impact, and ways of its conservation. Staff members of the IMB have also been involved in the international projects supported by the Asia-Pacific Network for Global Change Research (APN) [2]. We recently presented a synthesis of data and original research findings on coral reef modifications, biota of the intertidal zone, meiobenthic communities, species richness of rare groups of animals and economically important mollusks as a monograph [3], and here we provide a brief overview of some biodiversity problems in Vietnam.
II. Biodiversity of the intertidal zone
Recently, E.E. Kostina et al. [4] summarized in details all available data about the biodiversity of the intertidal zones of Vietnam, from the Gulf of Thailand in the south to the Gulf of Tonkin in the north, including those published by earlier French workers, national Vietnamese studies and Russian researches from 1980–2005. According to their review, the macrobenthos of hard substrata is the richest in qualitative and quantitative composition, and the population of soft substrata is the poorest (Fig. 1). Some 1664 species, subspecies or varieties of macrobenthic organisms (278 plant species and 1386 animal species) are found, with red algae dominating among plants (154 species), and gastropod mollusks (334 species) and decapod crustaceans (275 species) prevailing among animals.
The intertidal zone is most sensitive to any human-induced changes, e.g., overexploitation of biological resources, tourism pressure and pollution. One of the important intertidal ecosystems are mangrove systems, one of the most complex and productive world ecosystems, providing crucial livelihood support to coastal communities, but they are being degraded and lost in most countries. In Vietnam, the principal threat to mangrove ecosystems is their conversion to aquaculture [5]. Vietnam lost 69% of its 269000 ha of mangrove forests held in 1980, with an estimated 77% of this loss due to aquaculture [6].
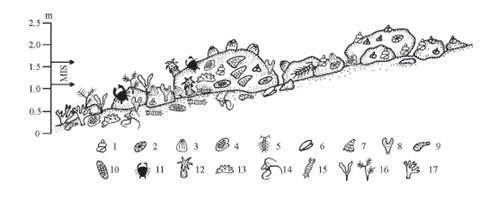
Fig. 1. Scheme of distribution of macrobenthos in the bouldery intertidal zone (from Nha Trang to Ro Bay). 1 – Gastropoda (mainly Littorinidae+Neritidae), 2 – Patellogastropoda, 3 – Cirripedia, 4 – Bivalvia, 5– Isopoda, 6 – Capitulum mitella, 7 – Saccostrea scyphophilla, 8 – Spongia, 9 – Sipuncula, 10 – Polyplacophora, 11 – Decapoda, 12 – Actiniaria, 13 – Holothuroidea, 14 – Ophiuroidea, 15 – Stomatopoda, 16 – algae, 17 – Scleractinia. Vertical scale – range over depths 0. MIS – the middle intertidal subzone (after [4]).
A monitoring system should be implemented to watch changes in the intertidal biotic communuties and their biodiversity. As this approach requires deep taxonomic knowledge and expertise for correct identification of animal and plant species, a collaboration between national and overseas reseachers is strongly needed.
III. Biodiversity of corals and coral reefs
As a result of more than 30-year studies of the reef-building scleractinians of Vietnam, 358 corals species belonging to 80 genera were found (Fig. 2), of which 153 species in 28 genera were recorded for the first time in the area [7, 8]. The species diversity of Vietnam’s reefs consists mainly of the members of five families: Acroporidae (98 species), Faviidae (42 species), Fungiidae (32 species), Poritidae (31 species), and Dendrophylliidae (26 species), making up altogether 64% of the total scleractinian species composition. The species composition and high diversity of Vietnamese corals are closely related to the Indonesia-Polynesian center of origin of the coral faunas [7, 8]. The species composition and high diversity of the Vietnamese coral fauna, as well as its close similarity to the southwest Pacific coral fauna, allow one to refer it to the Indonesia–Polynesian center of origin of the coral faunas of the tropical Indo-Pacific. The entire Vietnam coast is one biogeographical unit and is a part of the Indo-Polynesian Province of the Indo-Pacific Area [9].
Based on various literature data, the survey of coral reefs in Vietnam during the last 15 years shows that the area of coral reefs has been reduced by 15-20%, mainly in coastal waters of the central part of Vietnam from Da Nang to Binh Thuan Province [2]. Along with the coral reef area reduction, the species richness also decreased. For example, the coverage of coral reefs in Bai Tien area (Nha Trang) was 30% (1984), with 60 species, and it reduced to 1% by 1998 and the number of species decreased to 30 [2]. Other organisms diversity and abundance also reduced significantly. At present, all countries in the South China Sea have degraded reefs, from 95% in Hainan Island to an unknown level in Vietnam, and sustainable use, protection and restoration of Vietnamese coral reefs are most important problems of the coastal zone management [2].
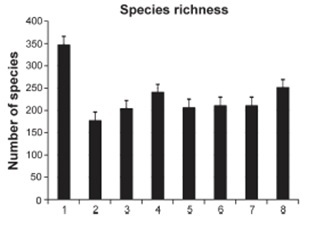
Fig. 2. The species richness of scleractinian species in different regions of Vietnam. 1 – total number of species (347), 2 – Gulf of Tonkin (177), 3 – Central Vietnam (204), 4 – South Vietnam (240), 5 – Thu Island (206), 6 – Con Dao Islands (210), 7 – Spratly Island (211), 8 – Gulf of Siam (251) (after [9]).
IV. Biodiversity of bivalve mollusks
The South China Sea has the most diverse bivalve fauna in the world from 1200–1500 species. Regional species richness of bivalve mollusks in the western sea area varies from 344, at Singapore in the south, to 822, southern mainland China in the north, more of an indication of sampling efforts and the state of knowledge of the fauna than real biodiversity gradients [10]. Vietnam is among the biodiversity-rich areas, and bivalve species richness of Vietnamese is 815 (Fig. 3).
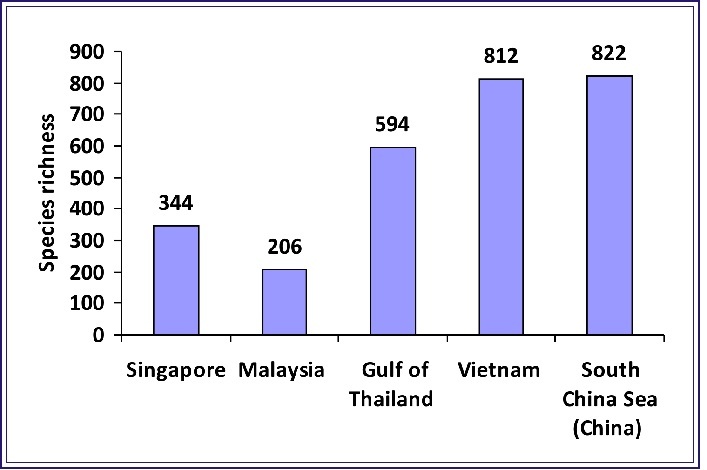
Fig.3. Species richness of bivalve mollusks in major regions of the western South China Sea (Singapore, Malaysia, the Gulf of Thailand, Vietnam and Chinese (mainland) part of the South China Sea).
Problems in studying the region’s bivalve biodiversity include inflated synonymy, a lack of trained, regionally-based taxonomists, good voucher collections, and a lack of funding for taxonomy.
V. Threats to marine biodiversity
Threats to marine biodiversity, including molluscan faunas, in the South China Sea are habitat degradation, fragmentation and loss -- especially mangrove forest destruction, loss of coral reefs, change in the landscape mosaic of wetland, estuary, and sand and mud flats; global climate change including sea level rise, storm events, rainfall pattern changes, and warming of the coastal ocean; effects of fishing and other forms of overexploitation, pollution and marine litter; species introduction/invasions, physical alterations of coasts, and tourism [11; 12; et al.].
Continued warming through the twenty-first century is inevitable and will likely have widespread ecological impacts [13]. In Vietnam, the Red (Song Hong) and Mekong rivers discharge into the sea, and the drainage systemns of these two transboundary rivers cover parts of six countries, and their water and sediment discharges greatly influence the coastal seas of Vietnam. The impact of human activity includes changes in the quality of the coastal and marine environments due to the increased use and accumulation of pollutants, and the loss of habitat. These impacts have resulted in the increasing unpredictability and severity of coastal problems, such as floods, erosion, sedimentation, saltwater intrusion, environmental pollution, and the degradation of ecosystems, with an accompanying decrease in biodiversity and fishery productivity [14].
Consolidated data of Vietnamese and Russian researchers on biodiversity and coastal zone management can be used in interpretations of ecosystem changes and for development of recommendations for local/national decision-makers.
1. B.W. Hoeksema, “Delineation of the Indo-Malayan centre of maximum marine biodiversity: the Coral Triangle” in Biogeography, Time, and Place: Distributions, Barriers, and Islands. W. Renema, Ed. Heidelberg, etc.: Springer, 2007, pp. 117-178.
2. K.A. Lutaenko, K.-S. Choi and Thái Ngọc Chiến, Coastal Marine Biodiversity of Vietnam: Regional and Local Challenges and Coastal Zone Management for Sustainable Development: Final Report for APN Project ARCP2011-10CMY-Lutaenko. Kobe: APN, 2012, unpublished.
3. A.V. Adrianov and K.A. Lutaenko, Eds., Biodiversity of the Western Part of the South China Sea, Vladivostok: Dalnauka, 2016.
4. E.E. Kostina, A.P. Tsurpalo and V.V. Gulbin, “The species composition and distribution of macrobenthic communities in the intertidal zone of Vietnam,” in Biodiversity of the Western Part of the South China Sea, A.V. Adrianov and K.A. Lutaenko, Eds. Vladivostok: Dalnauka, 2016, pp. 139-278.
5. S.E. Orchard, L.C. Stringer and C.H. Quinn, “Impacts of aquaculture on social networks in the mangrove systems of northern Vietnam,” Ocean Coast. Management, vol. 114, pp. 1-10, May 2015.
6. S. Hamilton, “Assessing the role of commercial aquaculture in displacing mangrove forest,” Bull. Mar. Sci., vol. 89, pp. 585-601, 2013.
7. Yu.Ya. Latypov, Coral Reefs of Vietnam, Moscow: Nauka, 2007.
8. Yu.Ya. Latypov, “Scleractinian corals and reefs of Vietnam as a part of the Pacific reef ecosystem,” Open J. Mar. Sci., vol. 1, pp. 50-68.
9. Yu.Ya. Latypov, “Reef-bulding corals of Vietnam as a part of the Indo-Pacific reef ecosystem,” in Biodiversity of the Western Part of the South China Sea, A.V. Adrianov and K.A. Lutaenko, Eds. Vladivostok: Dalnauka, 2016, pp. 111-138.
10. K.A. Lutaenko, “Biodiversity of bivalve mollusks in the western South China Sea: an overview”, in Biodiversity of the Western Part of the South China Sea, A.V. Adrianov and K.A. Lutaenko, Eds. Vladivostok: Dalnauka, 2016, pp. 315-384.
11. J.S. Gray, “Marine biodiversity: patterns, threats and conservation needs,” Biodiv. Conserv., vol. 6, pp. 153-175, 1997.
12. K.A. Lutaenko and N.C. Thái, “Coastal marine biodiversity of Vietnam: state and current problems,” APN Sci. Bull., N 4, pp. 18-22, 2014.
13. M.C. Serreze, “Understanding recent climate change,” Conserv. Biol., vol. 24, pp. 10-17, 2009.
14. Tran Duc Thanh, Saito Y., Dinh Van Huy, Van Lap Nguyen, Thi Kim Oanh Ta and M. Tateishi, “Regimes of human and climate impacts on coastal changes in Vietnam,” Reg. Environ. Change, vol. 4. pp. 49-62, 2004.







