BISAC NAT010000 Ecology
BISAC NAT045050 Ecosystems & Habitats / Coastal Regions & Shorelines
BISAC NAT025000 Ecosystems & Habitats / Oceans & Seas
BISAC NAT045030 Ecosystems & Habitats / Polar Regions
BISAC SCI081000 Earth Sciences / Hydrology
BISAC SCI092000 Global Warming & Climate Change
BISAC SCI020000 Life Sciences / Ecology
BISAC SCI039000 Life Sciences / Marine Biology
BISAC SOC053000 Regional Studies
BISAC TEC060000 Marine & Naval
Mass bleaching and subsequent mortality of scleractinian corals in response to elevated seawater temperatures has been considered as one of the most impacts of global climate change. Three extensive coral bleaching events in the Andaman Sea were reported, in the years 1991, 1995 and 2010. Studies on survival of coral colonies, coral recruitment and community structure of coral reef associated macrofauna would predict the trends for coral recovery from the impacts of coral bleaching events. The present study aimed to examine the status of coral communities, density of coral recruits and coral reef associated macrofauna at nearshore coral reefs in Phangnga Province, the Andaman Sea following the 2010 coral bleaching event. The dead coral cover was high (>50%) while the live coral cover was in the range of 13-21%. There was high diversity of coral recruits on natural substrates. The average densities of macrobenthic fauna varied from 1.9 to 2.6 individuals.m-2, with significant differences among study sites. The dominant macrobenthic species were a soft coral (Lobophytum sp.), a sea star (Linckia laevigata) and a sea urchin (Echinostrephus molaris). Coral recovery at these coral reefs would be possible but local anthropogenic stressors must be overwhelmingly reduced in order to enhance coral reef resilience. The long-term monitoring programs in the Andaman Sea are required for decision makers to support their adaptive management approaches.
coral reef, monitoring, climate change, recovery, Andaman Sea.
- INTRODUCTION
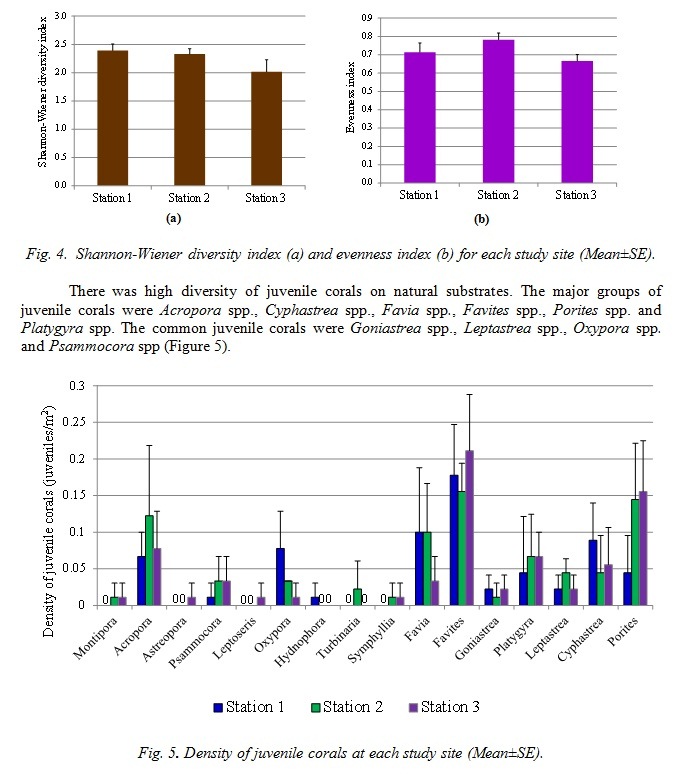
Mass coral bleaching and subsequent mortality in response to thermal anomalies has been recognized as one of the most severe impacts of global climate change [1]. Coral bleaching events have been reported with increasing frequency on coral communities around the world [2]. There have been three documented incidences of extensive coral bleaching in the Andaman Sea, in 1991, 1995 and 2010. The bleaching events were recorded in May at the time of maximum seasonal sea surface temperature (SST), with the maximum SST 32.5°C in 2010 [3, 4, 5]. The data on survival of coral colonies and their recruitment are needed to predict the trends for coral recovery following the coral bleaching events and to explain spatial and temporal variations in coral community structure [6, 7]. The impacts of coral declines on populations of reef associated macrofauna are also very important research aspects. This study aimed to quantitatively examine the status of coral communities, the abundance of juvenile corals and reef associated macrofauna at Hat Thai Mueang, Phangnga Province, the Andaman Sea following the 2010 coral bleaching event.
- MATERIAL AND METHODS
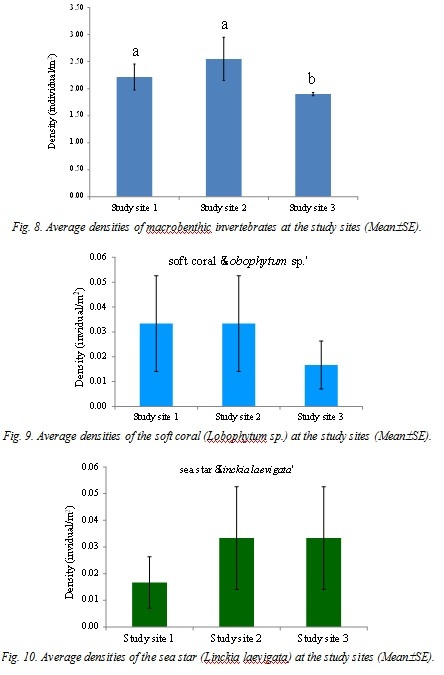
The study sites were located at Hat Thai Mueang, in the Andaman Sea. Three stations were set up in 2013 as the study sites, i.e. Station 1: 8°26'23.92"N, 98°13'27.70"E; Station 2: 8°26'10.17"N, 98°13'19.72"E; Station 3: 8°25'56.04" N E 98°13'20.37"E (Figure 1). The coral communities were found at about 4-5 m in depth. At each study site, live coral cover was recorded in three permanent belt-transects of 50x1 m2 and scleractinian corals (>5 cm diameter) were identified to genera level. The number of visible juvenile corals (1-5 cm diameter) was measured in these three permanent belt-transects. All juvenile corals were identified to genus. The numbers of each macro-invertebrate species found on the coral community were also recorded and counted in these permanent belt-transects and they were identified to a species level, if possible. The Shannon-Wiener index and Evenness index were calculated based on number of individuals for each study site. The data of a soft coral (Lobophytum sp.), sea star (Linckia laevigata) and a sea urchin (Echinostrephus molaris) densities were tested for normality and homogeneity of variances and were compared the influence of station by one-way ANOVA. Where significant differences were established. Tukey HSD was employed to determine which station(s) differed.
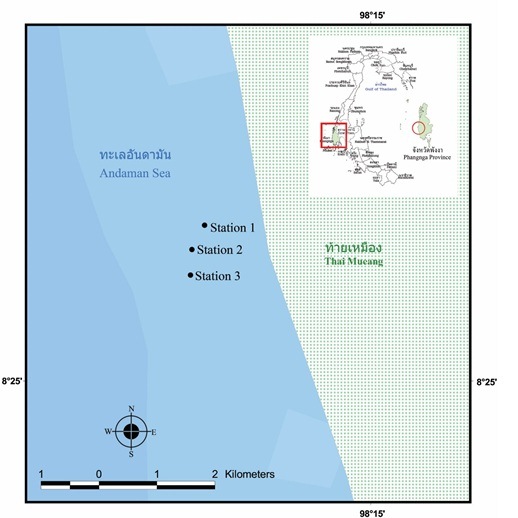
Fig. 1. Three study sites at Hat Thai Mueang, Phangnga Province, the Andaman Sea.
- RESULTS
The percentages of live coral cover were in the range of 13-21 while the range of dead coral cover was much higher (49-58%). The live coral cover following the 2010 coral bleaching event was not significantly different among the three study sites (Figure 2, ANOVA, p<0.05).
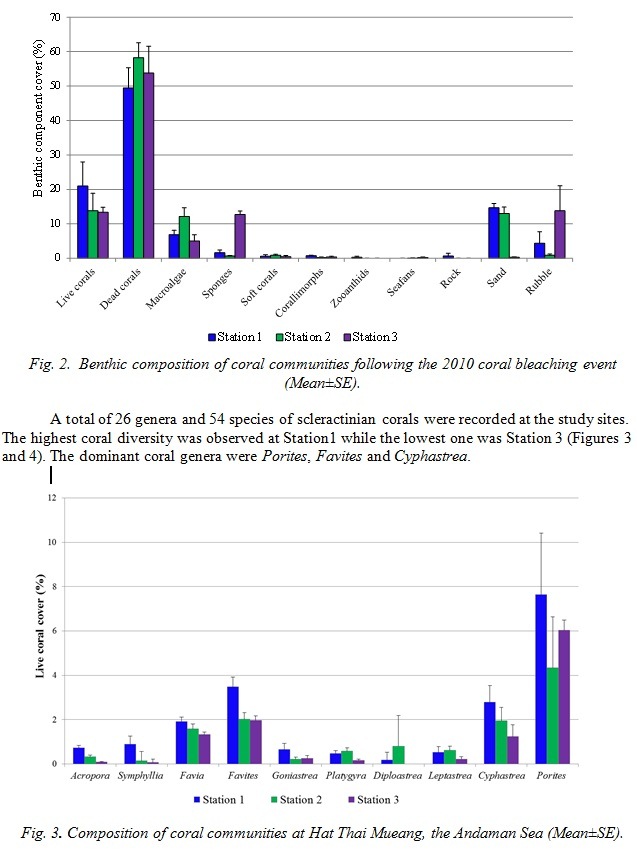


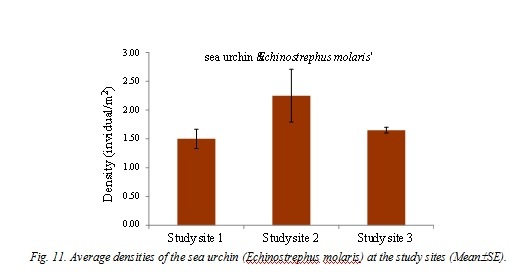
- DISCUSSION
Many dead coral colonies were observed at Haad Thai Muang during the 2010 coral bleaching event but there were relatively high diversity of juvenile corals following the coral bleaching. Therefore recovery of these coral communities would be possible. However, some coral communities in the Andaman Sea, such as Mu Ko Surin, have higher juvenile coral densities [8]. Population ecology of several microbenthic invertebrates on coral communities at Haad Thai Muang should be investigated to monitor the impacts of global climate change on coral reef-associated organisms. As no practical steps can be taken at the local level to prevent the thermal coral bleaching events, reducing anthropogenic disturbances such as water pollution, overfishing and heavy sedimentation is feasible and essential in order to maintain ecological balance of macroalgal cover and enhance coral reef resilience to climate change [9]. The long-term monitoring programs in the Andaman Sea have to be established in order to make adaptive management possible [10].
- ACKNOWLEDGMENTS
We are most grateful to staff of the Khao Lampi - Hat Thai Mueang National Park and Marine Biodiversity Research Group, Faculty of Science, Ramkhamhaeng University for their field work assistance. This research was funded by a budget for research promotion from the Ramkhamhaeng University.
1. O. Hoegh-Guldberg, J.F. Bruno, “The impact of climate change on the world’s marine ecosystems,” Science, vol. 328, pp. 1523-1528, 2010.
2. P.W. Glynn, “Coral reef bleaching: facts, hypotheses and implications,” Glob. Change Biol., vol. 2, pp. 495-509, 1996.
3. B.E. Brown, “BECoral bleaching: causes and consequences,” Coral Reefs, vol. 16, S129-S138, 1997.
4. T. Yeemin, C. Saenghaisuk, W. Klinthong, M. Yucharoen, M. Sutthacheep. “Impact of the 2010 coral bleaching event on survival of juvenile coral colonies in the Similan Islands, on the Andaman Sea coast of Thailand,” Phuket Mar. Biol. Cent. Res. Bull., vol. 71, pp. 93-102, 2012.
5. M. Sutthacheep, S. Pengsakun, W. Samsuvan, T. Yeemin. “Status of coral communities at Hat Tthai Mueang, Phangnga Province following the 2010 coral bleaching event,” [Proceedings of the 39th Science and Technology of Thailand, pp. 763-766, 2013].
6. J.H. Connell, T.P. Hughes, C.C. Wallace, “A 30-years study of coral abundance, recruitment, and disturbance at several scales in space and time,” Ecol. Monogr. vol. 67, pp. 461-488, 1997.
7. N. Knowlton, “The future of coral reefs,” Proc. Natl. Acad. Sci. U. S. A. vol. 98, pp. 5419-5425, 2001.
8. M. Yucharoen, T. Yeemin, B.E. Casareto, Y. Suzuki, W. Samsuvan, K. Sangmanee, W. Klinthong, S. Pengsakun, M. Sutthacheep. “Abundance, composition and growth rate of coral recruits on dead corals following the 2010 bleaching event at Mu Ko Surin, the Andaman Sea,” Ocean Sci. J. vol. 50, pp. 307-315, 2015.
9. J.A. Maynard, P.A. Marshall, J.E. Johnson, S. Harman. “Building resilience into practical conservation: identifying local management responses to global climate change in the southern Great Barrier Reef,” Coral Reefs, vol. 29, pp 381 - 391, 2010.
10. T. Yeemin, V. Mantachitra, S. Plathong, P. Nuclear, W. Klinthong, M. Sutthacheep. “Impacts of coral bleaching, recovery and management in Thailand,” [Proceedings of the 12th International Coral Reef Symposium, Australia, 5 pp., 2012].







